Candidates should be able to:
(a) *describe ionic (electrovalent) bonding, as in sodium chloride and magnesium oxide, including the use of ‘dot-and-cross’ diagrams
(b) *describe, including the use of ‘dot-and-cross’ diagrams,
(i) covalent bonding, as in hydrogen, oxygen, chlorine, hydrogen chloride, carbon dioxide, methane, ethene
(ii) co-ordinate (dative covalent) bonding, as in the formation of the ammonium ion and in the Al2Cl6 molecule
(c) *explain the shapes of, and bond angles in, molecules by using the qualitative model of electron-pair repulsion (including lone pairs), using as simple examples: BF3 (trigonal), CO2 (linear),CH4 (tetrahedral), NH3(pyramidal), H2O (non-linear), SF6 (octahedral)
(d) *describe covalent bonding in terms of orbital overlap, giving σ and π bonds
(e) *explain the shape of, and bond angles in, the ethane, ethene molecules in terms of σ
and π bonds (see also Section10.1)
(f) predict the shapes of, and bond angles in, molecules analogous to those specified in (c) and (e)
(g) describe hydrogen bonding, using ammonia and water as simple examples of molecules containing N-H and O-H groups
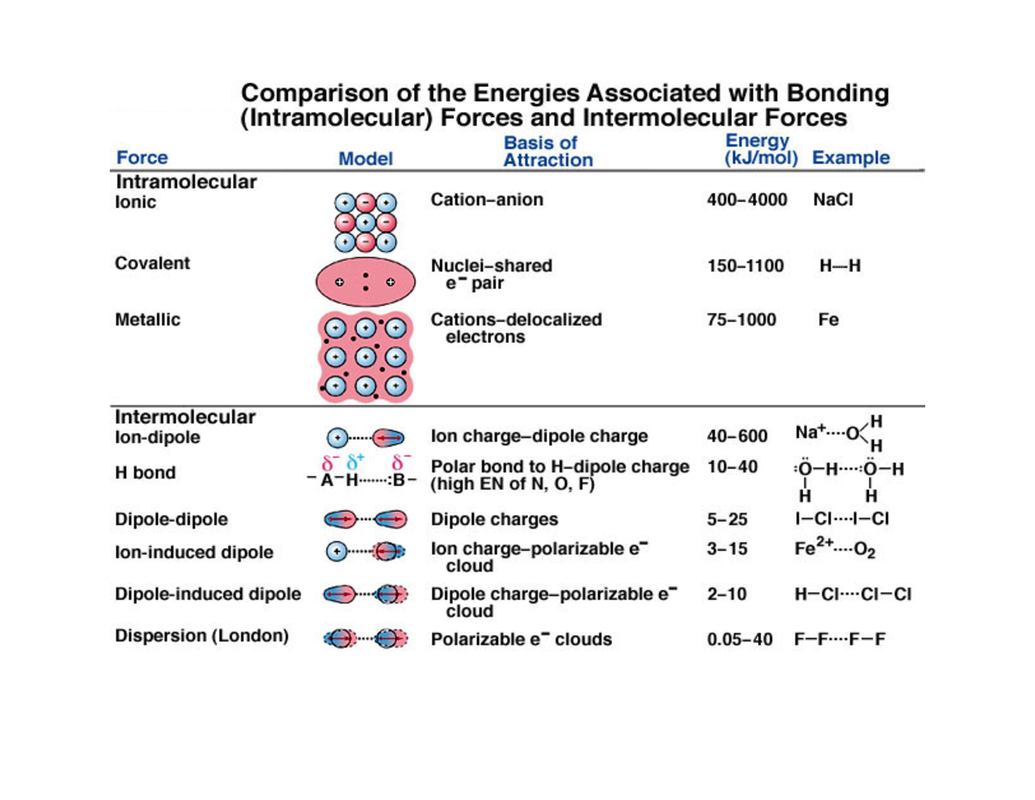
(h) explain the terms bond energy, bond length and bond polarity and use them to compare the reactivities of covalent bonds (see also 5b(ii))
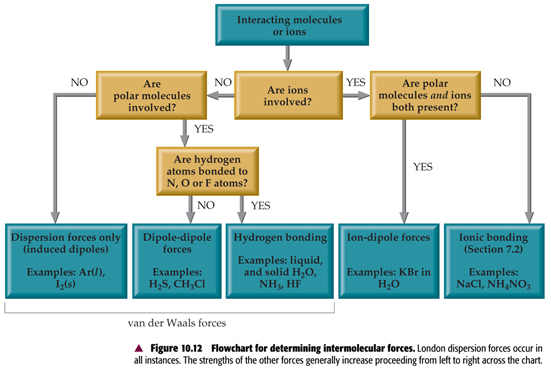
(i) *describe intermolecular forces (van der Waals’ forces), based on permanent and induced dipoles, as in CHCl 3(l) ; Br2(l) and the liquid noble gases
(j) describe metallic bonding in terms of a lattice of positive ions surrounded by mobile electrons
(k) *describe, interpret and/or predict the effect of different types of bonding (ionic bonding, covalent bonding, hydrogen bonding, other intermolecular interactions, metallic bonding) on the physical properties of substances
(l) deduce the type of bonding present from given information
(m) show understanding of chemical reactions in terms of energy transfers associated with the breaking and making of chemical bonds
(a) *describe ionic (electrovalent) bonding, as in sodium chloride and magnesium oxide, including the use of ‘dot-and-cross’ diagrams
(b) *describe, including the use of ‘dot-and-cross’ diagrams,
(i) covalent bonding, as in hydrogen, oxygen, chlorine, hydrogen chloride, carbon dioxide, methane, ethene
(ii) co-ordinate (dative covalent) bonding, as in the formation of the ammonium ion and in the Al2Cl6 molecule
(c) *explain the shapes of, and bond angles in, molecules by using the qualitative model of electron-pair repulsion (including lone pairs), using as simple examples: BF3 (trigonal), CO2 (linear),CH4 (tetrahedral), NH3(pyramidal), H2O (non-linear), SF6 (octahedral)
(d) *describe covalent bonding in terms of orbital overlap, giving σ and π bonds
(e) *explain the shape of, and bond angles in, the ethane, ethene molecules in terms of σ
and π bonds (see also Section10.1)
(f) predict the shapes of, and bond angles in, molecules analogous to those specified in (c) and (e)
(g) describe hydrogen bonding, using ammonia and water as simple examples of molecules containing N-H and O-H groups
(h) explain the terms bond energy, bond length and bond polarity and use them to compare the reactivities of covalent bonds (see also 5b(ii))
(i) *describe intermolecular forces (van der Waals’ forces), based on permanent and induced dipoles, as in CHCl 3(l) ; Br2(l) and the liquid noble gases
(j) describe metallic bonding in terms of a lattice of positive ions surrounded by mobile electrons
(k) *describe, interpret and/or predict the effect of different types of bonding (ionic bonding, covalent bonding, hydrogen bonding, other intermolecular interactions, metallic bonding) on the physical properties of substances
(l) deduce the type of bonding present from given information
(m) show understanding of chemical reactions in terms of energy transfers associated with the breaking and making of chemical bonds