Candidates should be able to:
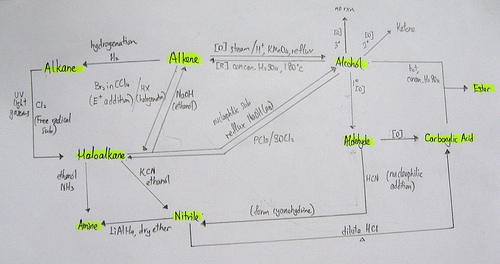
(a) *interpret, and use the general, structural, displayed and skeletal formulae of the following classes of compound:
(i) alkanes, and alkenes
(ii) halogenoalkanes
(iii) alcohols (including primary, secondary and tertiary)
(iv) aldehydes and ketones
(v) carboxylic acids, esters
(vi) amines (primary only), nitriles
[Candidates will be expected to recognise the shape of the
benzene ring when it is present in organic compounds. Knowledge of benzene or its compounds is not required for AS.]
(b) interpret, and use the following terminology associated with organic reactions:
(i) functional group
(ii) homolytic and heterolytic fission
(iii) free radical, initiation, propagation, termination
(iv) nucleophile, electrophile
(v) addition, substitution, elimination, hydrolysis
(vi) oxidation and reduction
[in equations for organic redox reactions, the symbols [O] and [H]
are acceptable]
(c) (i) *describe the shapes of the ethane, ethene
(ii) *predict the shapes of other related molecules
(d) *explain the shapes of the ethane, ethene molecules in terms of σ and π carbon-carbon bonds
(e) describe structural isomerism, and its division into chain, positional and functional group isomerism
(f) describe stereoisomerism, and its division into geometrical
(cis-trans) and optical isomerism
(g) describe cis-trans isomerism in alkenes, and explain its origin in terms of restricted rotation due to the presence of π bonds
(h) *explain what is meant by a chiral centre and that such a centre gives rise to optical isomerism
(i) identify chiral centres and/or cis-trans isomerism in a molecule of given structural formula
(j) deduce the possible isomers for an organic molecule of known molecular formula
(k) deduce the molecular formula of a compound, given its structural, displayed or skeletal formula (see Section 1)
(a) *interpret, and use the general, structural, displayed and skeletal formulae of the following classes of compound:
(i) alkanes, and alkenes
(ii) halogenoalkanes
(iii) alcohols (including primary, secondary and tertiary)
(iv) aldehydes and ketones
(v) carboxylic acids, esters
(vi) amines (primary only), nitriles
[Candidates will be expected to recognise the shape of the
benzene ring when it is present in organic compounds. Knowledge of benzene or its compounds is not required for AS.]
(b) interpret, and use the following terminology associated with organic reactions:
(i) functional group
(ii) homolytic and heterolytic fission
(iii) free radical, initiation, propagation, termination
(iv) nucleophile, electrophile
(v) addition, substitution, elimination, hydrolysis
(vi) oxidation and reduction
[in equations for organic redox reactions, the symbols [O] and [H]
are acceptable]
(c) (i) *describe the shapes of the ethane, ethene
(ii) *predict the shapes of other related molecules
(d) *explain the shapes of the ethane, ethene molecules in terms of σ and π carbon-carbon bonds
(e) describe structural isomerism, and its division into chain, positional and functional group isomerism
(f) describe stereoisomerism, and its division into geometrical
(cis-trans) and optical isomerism
(g) describe cis-trans isomerism in alkenes, and explain its origin in terms of restricted rotation due to the presence of π bonds
(h) *explain what is meant by a chiral centre and that such a centre gives rise to optical isomerism
(i) identify chiral centres and/or cis-trans isomerism in a molecule of given structural formula
(j) deduce the possible isomers for an organic molecule of known molecular formula
(k) deduce the molecular formula of a compound, given its structural, displayed or skeletal formula (see Section 1)
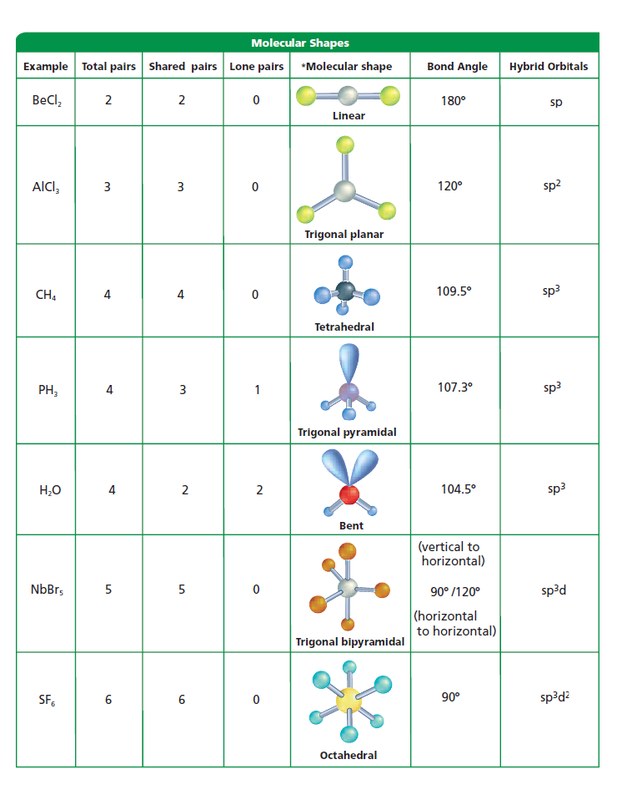
Carbon always forms four bonds. Carbon has a ground state configuration of 1s2 2s2 2px 2py, so it has 2 unpaired electrons in its outer p orbital. In order for Carbon to bond with four other atoms it must change its electron configuration to a higher energy excited state. This requires a release of energy to create a more stable compound. It will now become 1s2 2s1 2px 2py 2pz. Notice there a 4 unpaired electrons in the s and p orbitals, as s should have 2 electrons and the p orbital should have 6. The resulting orbitals will be hybridized.
Single bonds are sp3 hybrid orbitals with a tetrahedral geometry.
Double bonds are sp2 hybridized with 3 equivalent sp2 hybrid orbitals with planar geometry and one unhybridized p orbital.
Triple bonds are sp hybrid orbitals with 2 sp hybrid orbitals with linear geometry and two unhybridized p orbitals. These are the strongest bonds with the shortest bond length.
Nitrogen hybridizes to form four sp3 orbitals, exactly as carbon does, see the PH3 molecule below.
Oxygen and Boron will also hybridize. Oxygen will form two bonds Boron can form three bonds.
Single bonds are sp3 hybrid orbitals with a tetrahedral geometry.
Double bonds are sp2 hybridized with 3 equivalent sp2 hybrid orbitals with planar geometry and one unhybridized p orbital.
Triple bonds are sp hybrid orbitals with 2 sp hybrid orbitals with linear geometry and two unhybridized p orbitals. These are the strongest bonds with the shortest bond length.
Nitrogen hybridizes to form four sp3 orbitals, exactly as carbon does, see the PH3 molecule below.
Oxygen and Boron will also hybridize. Oxygen will form two bonds Boron can form three bonds.