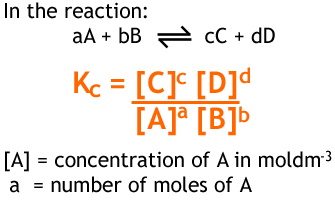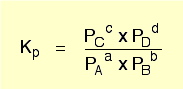Candidates should be able to:
(a) *explain, in terms of rates of the forward and reverse reactions,
what is meant by a reversible reaction and dynamic equilibrium
(b) *state Le Chatelier’s Principle and apply it to deduce qualitatively
(from appropriate information) the effects of changes in temperature, concentration or pressure, on a system at equilibrium
(c) deduce whether changes in concentration, pressure or temperature or the presence of a catalyst affect the value of the equilibrium constant for a reaction
(d) deduce expressions for equilibrium constants in terms of concentrations, Kc, and partial pressures, Kp
[treatment of the relationship between Kp and Kc is not required]
(e) calculate the values of equilibrium constants in terms of concentrations or partial pressures from appropriate data
(f) calculate the quantities present at equilibrium, given appropriate data (such calculations will not require the solving of quadratic equations)
(g) describe and explain the conditions used in the Haber process and the Contact process, as examples of the importance of an understanding of chemical equilibrium in the chemical industry (see also Section 9.6)
(h) show understanding of, and use the Brønsted-Lowry theory of acids and bases
(i) explain qualitatively the differences in behaviour between strong and weak acids and bases and the pH values of their aqueous solutions in terms of the extent of dissociation
Chapter 8 “Equilibrium”
8.1
Forward reaction – one where the reactants yields products and it is irreversible.
Reversible reaction – one where the products can react together to reform the reactants.
Dynamic equilibrium – molecules of reactants are being converted to products at the same rate as products are being converted to reactants.
Closed system – none of the reactants or products can escape into the surroundings.
Open system – reactants or products can escape into the surroundings
8.2
Le Chatelier’s principle – when any of the conditions affecting the position of equilibrium are changed, the position of that equilibrium shifts to minimize the change.
If reactants are increased the equilibrium moves to the right and more products are formed until equilibrium is again reached
If products are increased the equilibrium moves to the left and more reactants are formed until equilibrium is again reached
Change in pressure would affect gases only. By increasing pressure a shift would occur in the direction of less gas molecules. By decreasing pressure a shift would occur in the direction of more gas molecules. If there are equal molecules of gases on both sides then pressure would not be affected by a change in pressure.
Endothermic reactions are favored with an increase in temperature and the reaction shifts to the right. Decreasing temperature would shift it to the left.
Exothermic reactions are favored with a decrease in temperature and the reaction shifts to the right. Increasing temperatures would shift it to the left.
Catalysts speed up the time taken to reach equilibrium but have no effect on the position of equilibrium once it is reached.
8.3
see kc picture below
Does not include solids or pure water in its liquid state. A change in temperature would affect the equilibrium constant based on whether it is an endo or exothermic reactions. Refer to notation above. However, changes in concentration, pressure of presence of a catalyst do not affect the value of the constant.
8.4
Partial pressure – the pressure exerted by any one gas in a mixture. In other words the total pressure of a gas equals the sum of the partial pressures of all the gases present. Units of pressure are in pascals.
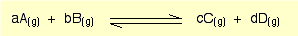
see kp picture below
8.5 Le Chatelier’s principle is used to increase the volumes of ammonia using iron as a catalyst in the haber process and sulfuric acid using di-vanadium pentoxide in the contact process. Remember enzymes are also catalysts. Haber process if at too high of a temperature will have a low yield but if at low temperature will have high yield.
8.6
Acid – a substance that neutralizes a base.
Base – a substance that neutralizes an acid
Acid + base à salt + water
Alkali – a base that is soluble in water.
Bronsted-lowry acid is a proton donor
Bronsted-lowry base is a proton acceptor
Amphoteric – a substance that can be either an acid or base, such as water
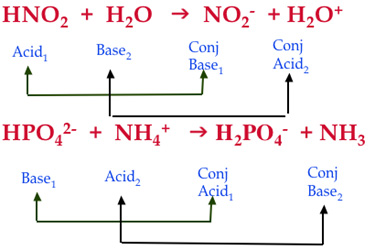
Conjugate pair – (see picture below) reactants linked to a product by the transfer or a proton
Strong acids – acids which dissociate almost completely in solution such as hydrochloric, sulfuric and nitric acid
Weak acids – acids which are only partially dissociated in solution such as hydrocyanic acid, hydrogen sulfide and carbonic acid
Strong bases – bases which dissociate almost completely in solution such as group I metal hydroxides.
Weak bases - bases which dissociate to only a small extent in solution such as ammonia
(a) *explain, in terms of rates of the forward and reverse reactions,
what is meant by a reversible reaction and dynamic equilibrium
(b) *state Le Chatelier’s Principle and apply it to deduce qualitatively
(from appropriate information) the effects of changes in temperature, concentration or pressure, on a system at equilibrium
(c) deduce whether changes in concentration, pressure or temperature or the presence of a catalyst affect the value of the equilibrium constant for a reaction
(d) deduce expressions for equilibrium constants in terms of concentrations, Kc, and partial pressures, Kp
[treatment of the relationship between Kp and Kc is not required]
(e) calculate the values of equilibrium constants in terms of concentrations or partial pressures from appropriate data
(f) calculate the quantities present at equilibrium, given appropriate data (such calculations will not require the solving of quadratic equations)
(g) describe and explain the conditions used in the Haber process and the Contact process, as examples of the importance of an understanding of chemical equilibrium in the chemical industry (see also Section 9.6)
(h) show understanding of, and use the Brønsted-Lowry theory of acids and bases
(i) explain qualitatively the differences in behaviour between strong and weak acids and bases and the pH values of their aqueous solutions in terms of the extent of dissociation
Chapter 8 “Equilibrium”
8.1
Forward reaction – one where the reactants yields products and it is irreversible.
Reversible reaction – one where the products can react together to reform the reactants.
Dynamic equilibrium – molecules of reactants are being converted to products at the same rate as products are being converted to reactants.
Closed system – none of the reactants or products can escape into the surroundings.
Open system – reactants or products can escape into the surroundings
8.2
Le Chatelier’s principle – when any of the conditions affecting the position of equilibrium are changed, the position of that equilibrium shifts to minimize the change.
If reactants are increased the equilibrium moves to the right and more products are formed until equilibrium is again reached
If products are increased the equilibrium moves to the left and more reactants are formed until equilibrium is again reached
Change in pressure would affect gases only. By increasing pressure a shift would occur in the direction of less gas molecules. By decreasing pressure a shift would occur in the direction of more gas molecules. If there are equal molecules of gases on both sides then pressure would not be affected by a change in pressure.
Endothermic reactions are favored with an increase in temperature and the reaction shifts to the right. Decreasing temperature would shift it to the left.
Exothermic reactions are favored with a decrease in temperature and the reaction shifts to the right. Increasing temperatures would shift it to the left.
Catalysts speed up the time taken to reach equilibrium but have no effect on the position of equilibrium once it is reached.
8.3
see kc picture below
Does not include solids or pure water in its liquid state. A change in temperature would affect the equilibrium constant based on whether it is an endo or exothermic reactions. Refer to notation above. However, changes in concentration, pressure of presence of a catalyst do not affect the value of the constant.
8.4
Partial pressure – the pressure exerted by any one gas in a mixture. In other words the total pressure of a gas equals the sum of the partial pressures of all the gases present. Units of pressure are in pascals.
see kp picture below
8.5 Le Chatelier’s principle is used to increase the volumes of ammonia using iron as a catalyst in the haber process and sulfuric acid using di-vanadium pentoxide in the contact process. Remember enzymes are also catalysts. Haber process if at too high of a temperature will have a low yield but if at low temperature will have high yield.
8.6
Acid – a substance that neutralizes a base.
Base – a substance that neutralizes an acid
Acid + base à salt + water
Alkali – a base that is soluble in water.
Bronsted-lowry acid is a proton donor
Bronsted-lowry base is a proton acceptor
Amphoteric – a substance that can be either an acid or base, such as water
Conjugate pair – (see picture below) reactants linked to a product by the transfer or a proton
Strong acids – acids which dissociate almost completely in solution such as hydrochloric, sulfuric and nitric acid
Weak acids – acids which are only partially dissociated in solution such as hydrocyanic acid, hydrogen sulfide and carbonic acid
Strong bases – bases which dissociate almost completely in solution such as group I metal hydroxides.
Weak bases - bases which dissociate to only a small extent in solution such as ammonia