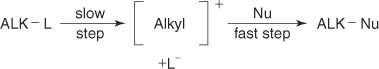Reaction Mechanisms:
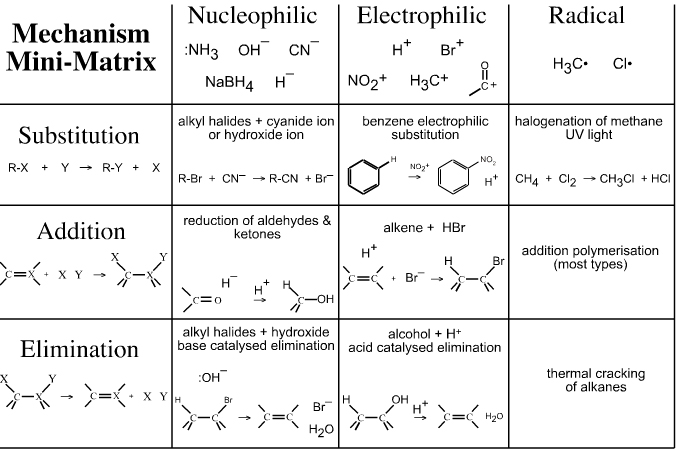
Two ways in which a covalent bond can break:
Homolytic - free radical is formed, where one electron stays with each fragment
Chlorination of Methane.
Heterolytic - polar compound is formed, where 2 electrons stay with one fragment
Carbon has a partial positive charge when bonded to O,N,F,Cl or Br.
Bonds are made when an electron rich reagent donates a pair of electrons to the electron deficient reagent.
Bonds are broken when either pair leaves with the electron pair.
The electron pair movement is designated with curved arrows, which shows the motion of the electrons.
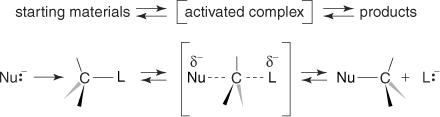
A Nucleophile (usually a base, carbon=carbon) is a reagent that is electron rich and can form a bond by donating a pair of electrons to an electron poor site. An Electrophile (usually an acid, alkyl halides) is electron poor and will accept electrons from the nucleophile.
Sn2 reactions - nucleophile and alkyl halide reaction occurs with complete inversion of steriochemistry at the chiral carbon center. Look for these compounds as nucleophiles: Hydride, Cyanide, Iodide, Hydroxide, Methoxide, Chloride, Acetate, Ammonia. Favored for primary and secondary substrates. Aprotic solvents are favored for this reaction: Hexane, Benzene, Diethyl Ether, Chloroform, Ethyl Acetate, Acetone, HMPA, DMF, DMSO. Rates of reaction: Alkyl halide > primary > secondary
Sn1 reactions - the substrate dissociates into a carbocation ( formed as an intermediate) in a slow reaction followed by a fast attack by the nucleophile. Favored for tertiary substrates. Protic solvents are favored for this reaction: Acetic Acid, tert-Butyl alcohol, Ethanol, Methanol, Formic Acid and Water.
Solvents can be polar or non-polar.
Polar solvents can be divided into protic or aprotic. A protic solvent is one that has a hydrogen atom bound to an oxygen or nitrogen. An aprotic solvent is one that does not form hydrogen bonds.
The Seven Organic Sequences:
1. CH3CH3 = Cl2/uv => CH3CH2Cl = reflux/NaOH(aq) => CH3CH2OH = K2Cr2O7/H+(aq) => CH3COOH
2. CH3CH2CH3 = heat/catalyst => CH3CH=CH2 = conc.HBr(aq) => CH3CHBrCH3 = NaOH(aq)/reflux =>CH3CHOHCH3 = CH3COCl / RTP => CH3COOCH(CH3)2
3. CH3CH3 = heat/catalyst => CH2=CH2 = HBr => CH3CH2Br = conc.NH3(aq / ethanol) => CH3CH2NH2 = CH3COCl / RTP => CH3CONHCH2CH3
4. CH3CH3 = heat/catalyst => CH2=CH2 = H2O/H3PO4 => CH3CH2OH = K2Cr2O7/ H+(aq) / distil => CH3CHO = HCN(aq) => CH3CH(OH)CN = H+ / reflux => CH3CH(OH)COOH = K2Cr2O7 / H+(aq) / reflux => CH3COCOOH
5. C6H6 = CH3Cl / AlCl3 => C6H5CH3 = KMnO4/H+(aq) => C6H5COOH = CH3OH/conc.H2SO4 => C6H5COOCH3
6. C6H6 = CH3Cl / AlCl3 => C6H5CH3 = Cl2 / uv => C6H5CH2Cl = conc.NH3(aq/ethanol) => C6H5CH2NH2
7. C6H6 =conc.HNO3 / H2SO4 / 60 C => C6H5NO2 = Sn / conc.HCl(aq) / reflux => C6H5NH2 = NaNO2 / HCl(aq) / 5 C => C6H5N2 + Cl = C6H5OH / NaOH (aq) / RTP => C6H5-N=N-C6H4OH
1. CH3CH3 = Cl2/uv => CH3CH2Cl = reflux/NaOH(aq) => CH3CH2OH = K2Cr2O7/H+(aq) => CH3COOH
2. CH3CH2CH3 = heat/catalyst => CH3CH=CH2 = conc.HBr(aq) => CH3CHBrCH3 = NaOH(aq)/reflux =>CH3CHOHCH3 = CH3COCl / RTP => CH3COOCH(CH3)2
3. CH3CH3 = heat/catalyst => CH2=CH2 = HBr => CH3CH2Br = conc.NH3(aq / ethanol) => CH3CH2NH2 = CH3COCl / RTP => CH3CONHCH2CH3
4. CH3CH3 = heat/catalyst => CH2=CH2 = H2O/H3PO4 => CH3CH2OH = K2Cr2O7/ H+(aq) / distil => CH3CHO = HCN(aq) => CH3CH(OH)CN = H+ / reflux => CH3CH(OH)COOH = K2Cr2O7 / H+(aq) / reflux => CH3COCOOH
5. C6H6 = CH3Cl / AlCl3 => C6H5CH3 = KMnO4/H+(aq) => C6H5COOH = CH3OH/conc.H2SO4 => C6H5COOCH3
6. C6H6 = CH3Cl / AlCl3 => C6H5CH3 = Cl2 / uv => C6H5CH2Cl = conc.NH3(aq/ethanol) => C6H5CH2NH2
7. C6H6 =conc.HNO3 / H2SO4 / 60 C => C6H5NO2 = Sn / conc.HCl(aq) / reflux => C6H5NH2 = NaNO2 / HCl(aq) / 5 C => C6H5N2 + Cl = C6H5OH / NaOH (aq) / RTP => C6H5-N=N-C6H4OH