Candidates should be able to:
(a) explain and use the terms: rate of reaction, activation energy, catalysis
(b) *explain qualitatively, in terms of collisions, the effect of concentration changes on the rate of a reaction
(c) *show understanding, including reference to the Boltzmann distribution, of what is meant by the term activation energy
(d) *explain qualitatively, in terms both of the Boltzmann distribution and of collision frequency, the effect of temperature change on the rate of a reaction
(e)*(i) explain that, in the presence of a catalyst, a reaction has a different mechanism, i.e. one of lower activation energy
(ii) interpret this catalytic effect in terms of the Boltzmann distribution
(f) describe enzymes as biological catalysts (proteins) which may have specific activity
The rate of a reaction = change in amount of reactants or products / time
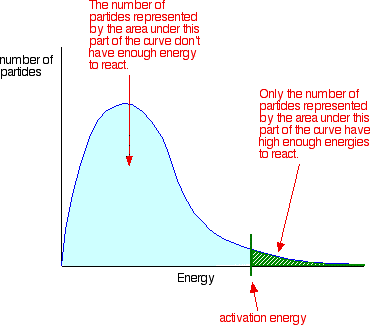
collision theory - states that in order to react with each other, particles must collide in the correct orientation and with sufficient energy. It can be deemed successful if there is enough energy to convert reactants into products.
a reaction will speed up if: the frequency of collisions increases and the proportion of particles with energy greater than the activation energy increases.
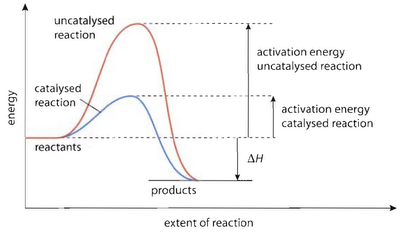
activation energy - the minimum energy needed for a successful collision to take place
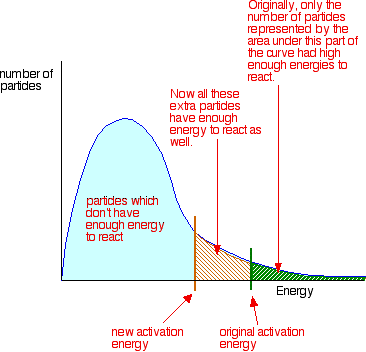
catalyst - lowers the activation energy needed by allowing the reaction to occur through an alternative mechanism
(a) explain and use the terms: rate of reaction, activation energy, catalysis
(b) *explain qualitatively, in terms of collisions, the effect of concentration changes on the rate of a reaction
(c) *show understanding, including reference to the Boltzmann distribution, of what is meant by the term activation energy
(d) *explain qualitatively, in terms both of the Boltzmann distribution and of collision frequency, the effect of temperature change on the rate of a reaction
(e)*(i) explain that, in the presence of a catalyst, a reaction has a different mechanism, i.e. one of lower activation energy
(ii) interpret this catalytic effect in terms of the Boltzmann distribution
(f) describe enzymes as biological catalysts (proteins) which may have specific activity
The rate of a reaction = change in amount of reactants or products / time
collision theory - states that in order to react with each other, particles must collide in the correct orientation and with sufficient energy. It can be deemed successful if there is enough energy to convert reactants into products.
a reaction will speed up if: the frequency of collisions increases and the proportion of particles with energy greater than the activation energy increases.
activation energy - the minimum energy needed for a successful collision to take place
catalyst - lowers the activation energy needed by allowing the reaction to occur through an alternative mechanism
The more concentrated the solution the greater the frequency of collisions and the faster the rate of the reaction.
Boltzmann distribution - the distribution in the amount of particle energy present in a reaction.
Boltzmann distribution - the distribution in the amount of particle energy present in a reaction.
Using a catalyst provides a lower activation energy so a greater proportion of molecules can now react.
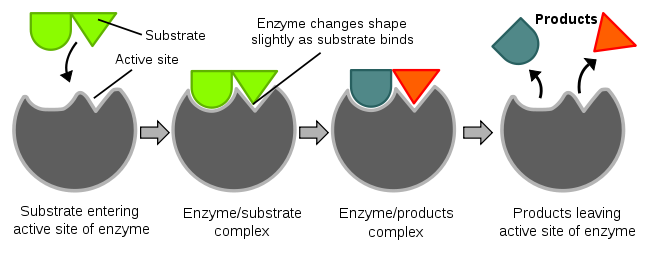
Enzymes are biological catalysts and will only catalyze one reaction involving one molecule or pairs of molecules. This is known as specificity. The enzyme has an active site which is a specific location that a molecule called a substrate fits into. This is known as the "lock and key model".
Enzymes loose their effectiveness and become denatured (loose their active site shape and substrate can no longer fit into it) above 45 degrees Celcius or 110 degrees Farenheit and at certain pH's.