In order to form bonds in Organic molecules a carbon will first hybridize to an excited-state configuration. A sigma bond is a bond that has a cylindrical shape about the bond axis. It can be formed when two s orbitals overlap such as a Hydrogen bonding with Hydrogen both of which have the electron configuration 1s1. It can also be formed when two p orbitals overlap on their axes such as when a Fluorine bonds with Fluorine both of which have the outer electron configuration 2p5.
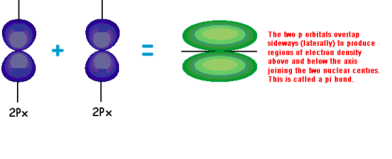
A pi bond is a bond that has electrons distributed above and below the bond axis. It is formed by the sideways overlap of two 2p orbitals. It will not be as strong as a sigma bond and occurs when two parallel orbitals are still available after a sigma bond has been formed.